
CHEBI:16618
| Name | 1-phosphatidyl-1D-myo-inositol 3,4,5-trisphosphate | 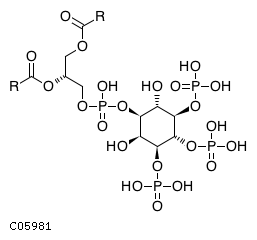 |
| Synonyms | 01,2-diacyl-sn-glycero-3-phospho-(1'-myo-inositol-3',4',5'-bisphosphate); 1-phosphatidyl-1d-myo-inositol 3,4,5-trisphosphate; 2,3-bis(alkanoyloxy)propyl (1s,2s,3r,4s,5s,6s)-2,6-dihydroxy-3,4,5-tris(phosphonatooxy)cyclohexyl phosphate; Phosphatidylinositol-3,4,5-trisphosphate; Pi(3,4,5)p3; Pip3; Pips; Ptdins(3,4,5)p3; Ptsins(3,4,5)p3; Ptsins-3,4,5-p3; | |
| Definition | NA | |
| Molecular Weight (Exact mass) | NA | |
| Molecular Formula | C11H20O22P4R2 | |
| SMILES | [H][C@@](COC([*])=O)(COP(O)(=O)O[C@@H]1[C@H](O)[C@H](OP(O)(O)=O)[C@@H](OP(O)(O)=O)[C@H](OP(O)(O)=O)[C@H]1O)OC([*])=O | |
| InChI | InChI=1S/C6H15O15P3/c7-1-2(8)5(20-23(13,14)15)6(21-24(16,17)18)3(9)4(1)19-22(10,11)12/h1-9H,(H2,10,11,12)(H2,13,14,15)(H2,16,17,18)/t1-,2+,3+,4-,5-,6-/m1/s1 | |
| InChI Key | MMWCIQZXVOZEGG-XJTPDSDZSA-N | |
| Crosslinking annotations | KEGG:C05981 | ChEBI:16618 | PubChem:8260 |
| Pathway ID | Pathway Name | Pathway Description (KEGG) |
| map00562 | Inositol phosphate metabolism | NA |
| map01100 | Metabolic pathways | NA |
| map01521 | EGFR tyrosine kinase inhibitor resistance | EGFR is a tyrosine kinase that participates in the regulation of cellular homeostasis. EGFR also serves as a stimulus for cancer growth. EGFR gene mutations and protein overexpression, both of which activate down- stream pathways, are associated with cancers, especially lung cancer. Several tyrosine kinase inhibitor (TKI) therapies against EGFR are currently administered and are initially effective in cancer patients who have EGFR mutations or aberrant activation of EGFR. However, the development of TKI resistance is common and results in the recurrence of tumors. Studies over the last decade have identified mechanisms that drive resistance to EGFR TKI treatment. Most outstanding mechanisms are: the secondary EGFR mutation (T790M), activation of alternative pathways (c-Met, HGF, AXL), aberrance of the downstream pathways (K-RAS mutations, loss of PTEN), impairment of the EGFR-TKIs-mediated apoptosis pathway (BCL2-like 11/BIM deletion polymorphism), histologic transformation, etc. |
| map04012 | ErbB signaling pathway | The ErbB family of receptor tyrosine kinases (RTKs) couples binding of extracellular growth factor ligands to intracellular signaling pathways regulating diverse biologic responses, including proliferation, differentiation, cell motility, and survival. Ligand binding to the four closely related members of this RTK family -epidermal growth factor receptor (EGFR, also known as ErbB-1 or HER1), ErbB-2 (HER2), ErbB-3 (HER3), and ErbB-4 (HER4)-induces the formation of receptor homo- and heterodimers and the activation of the intrinsic kinase domain, resulting in phosphorylation on specific tyrosine residues (pY) within the cytoplasmic tail. Signaling effectors containing binding pockets for pY-containing peptides are recruited to activated receptors and induce the various signaling pathways. The Shc- and/or Grb2-activated mitogen-activated protein kinase (MAPK) pathway is a common target downstream of all ErbB receptors. Similarly, the phosphatidylinositol-3-kinase (PI-3K) pathway is directly or indirectly activated by most ErbBs. Several cytoplasmic docking proteins appear to be recruited by specific ErbB receptors and less exploited by others. These include the adaptors Crk, Nck, the phospholipase C gamma (PLCgamma), the intracellular tyrosine kinase Src, or the Cbl E3 ubiquitin protein ligase. |
| map04014 | Ras signaling pathway | The Ras proteins are GTPases that function as molecular switches for signaling pathways regulating cell proliferation, survival, growth, migration, differentiation or cytoskeletal dynamism. Ras proteins transduce signals from extracellular growth factors by cycling between inactive GDP-bound and active GTP-bound states. The exchange of GTP for GDP on RAS is regulated by guanine nucleotide exchange factors (GEFs) and GTPase-activating proteins (GAPs). Activated RAS (RAS-GTP) regulates multiple cellular functions through effectors including Raf, phosphatidylinositol 3-kinase (PI3K) and Ral guanine nucleotide-dissociation stimulator (RALGDS). |
| map04062 | Chemokine signaling pathway | Inflammatory immune response requires the recruitment of leukocytes to the site of inflammation upon foreign insult. Chemokines are small chemoattractant peptides that provide directional cues for the cell trafficking and thus are vital for protective host response. In addition, chemokines regulate plethora of biological processes of hematopoietic cells to lead cellular activation, differentiation and survival.The chemokine signal is transduced by chemokine receptors (G-protein coupled receptors) expressed on the immune cells. After receptor activation, the alpha- and beta-gamma-subunits of G protein dissociate to activate diverse downstream pathways resulting in cellular polarization and actin reorganization. Various members of small GTPases are involved in this process. Induction of nitric oxide and production of reactive oxygen species are as well regulated by chemokine signal via calcium mobilization and diacylglycerol production. |
| map04068 | FoxO signaling pathway | The forkhead box O (FOXO) family of transcription factors regulates the expression of genes in cellular physiological events including apoptosis, cell-cycle control, glucose metabolism, oxidative stress resistance, and longevity. A central regulatory mechanism of FOXO proteins is phosphorylation by the serine-threonine kinase Akt/protein kinase B (Akt/PKB), downstream of phosphatidylinositol 3-kinase (PI3K), in response to insulin or several growth factors. Phosphorylation at three conserved residues results in the export of FOXO proteins from the nucleus to the cytoplasm, thereby decreasing expression of FOXO target genes. In contrast, the stress-activated c-Jun N-terminal kinase (JNK) and the energy sensing AMP-activated protein kinase (AMPK), upon oxidative and nutrient stress stimuli phosphorylate and activate FoxOs. Aside from PKB, JNK and AMPK, FOXOs are regulated by multiple players through several post-translational modifications, including phosphorylation, but also acetylation, methylation and ubiquitylation. |
| map04070 | Phosphatidylinositol signaling system | NA |
| map04071 | Sphingolipid signaling pathway | Sphingomyelin (SM) and its metabolic products are now known to have second messenger functions in a variety of cellular signaling pathways. Particularly, the sphingolipid metabolites, ceramide (Cer) and sphingosine-1-phosphate (S1P), have emerged as a new class of potent bioactive molecules. Ceramide can be generated de novo or by hydrolysis of membrane sphingomyelin by sphingomyelinase (SMase). Ceramide is subsequently metabolized by ceramidase to generate sphingosine (Sph) which in turn produces S1P through phosphorylation by sphingosine kinases 1 and 2 (SphK1, 2). Both ceramide and S1P regulate cellular responses to stress, with generally opposing effects. S1P functions as a growth and survival factor, acting as a ligand for a family of G protein-coupled receptors, whereas ceramide activates intrinsic and extrinsic apoptotic pathways through receptor-independent mechanisms. |
| map04072 | Phospholipase D signaling pathway | Phospholipase D (PLD) is an essential enzyme responsible for the production of the lipid second messenger phosphatidic acid (PA), which is involved in fundamental cellular processes, including membrane trafficking, actin cytoskeleton remodeling, cell proliferation and cell survival. PLD activity can be stimulated by a large number of cell surface receptors and is elaborately regulated by intracellular factors, including protein kinase C isoforms, small GTPases of the ARF, Rho and Ras families and the phosphoinositide, phosphatidylinositol 4,5-bisphosphate (PIP2). The PLD-produced PA activates signaling proteins and acts as a node within the membrane to which signaling proteins translocate. Several signaling proteins, including Raf-1 and mTOR, directly bind PA to mediate translocation or activation, respectively. |
| map04140 | Autophagy - animal | Autophagy (or macroautophagy) is a cellular catabolic pathway involving in protein degradation, organelle turnover, and non-selective breakdown of cytoplasmic components, which is evolutionarily conserved among eukaryotes and exquisitely regulated. This progress initiates with production of the autophagosome, a double-membrane intracellular structure of reticular origin that engulfs cytoplasmic contents and ultimately fuses with lysosomes for cargo degradation. Autophagy is regulated in response to extra- or intracellular stress and signals such as starvation, growth factor deprivation and ER stress. Constitutive level of autophagy plays an important role in cellular homeostasis and maintains quality control of essential cellular components. |
| map04144 | Endocytosis | Endocytosis is a mechanism for cells to remove ligands, nutrients, and plasma membrane (PM) proteins, and lipids from the cell surface, bringing them into the cell interior. Transmembrane proteins entering through clathrin-dependent endocytosis (CDE) have sequences in their cytoplasmic domains that bind to the APs (adaptor-related protein complexes) and enable their rapid removal from the PM. In addition to APs and clathrin, there are numerous accessory proteins including dynamin. Depending on the various proteins that enter the endosome membrane, these cargoes are sorted to distinct destinations. Some cargoes, such as nutrient receptors, are recycled back to the PM. Ubiquitylated membrane proteins, such as activated growth-factor receptors, are sorted into intraluminal vesicles and eventually end up in the lysosome lumen via multivesicular endosomes (MVEs). There are distinct mechanisms of clathrin-independent endocytosis (CIE) depending upon the cargo and the cell type. |
| map04150 | mTOR signaling pathway | The mammalian (mechanistic) target of rapamycin (mTOR) is a highly conserved serine/threonine protein kinase, which exists in two complexes termed mTOR complex 1 (mTORC1) and 2 (mTORC2). mTORC1 contains mTOR, Raptor, PRAS40, Deptor, mLST8, Tel2 and Tti1. mTORC1 is activated by the presence of growth factors, amino acids, energy status, stress and oxygen levels to regulate several biological processes, including lipid metabolism, autophagy, protein synthesis and ribosome biogenesis. On the other hand, mTORC2, which consists of mTOR, mSin1, Rictor, Protor, Deptor, mLST8, Tel2 and Tti1, responds to growth factors and controls cytoskeletal organization, metabolism and survival. |
| map04151 | PI3K-Akt signaling pathway | The phosphatidylinositol 3' -kinase(PI3K)-Akt signaling pathway is activated by many types of cellular stimuli or toxic insults and regulates fundamental cellular functions such as transcription, translation, proliferation, growth, and survival. The binding of growth factors to their receptor tyrosine kinase (RTK) or G protein-coupled receptors (GPCR) stimulates class Ia and Ib PI3K isoforms, respectively. PI3K catalyzes the production of phosphatidylinositol-3,4,5-triphosphate (PIP3) at the cell membrane. PIP3 in turn serves as a second messenger that helps to activate Akt. Once active, Akt can control key cellular processes by phosphorylating substrates involved in apoptosis, protein synthesis, metabolism, and cell cycle. |
| map04152 | AMPK signaling pathway | AMP-activated protein kinase (AMPK) is a serine threonine kinase that is highly conserved through evolution. AMPK system acts as a sensor of cellular energy status. It is activated by increases in the cellular AMP:ATP ratio caused by metabolic stresses that either interfere with ATP production (eg, deprivation for glucose or oxygen) or that accelerate ATP consumption (eg, muscle contraction). Several upstream kinases, including liver kinase B1 (LKB1), calcium/calmodulin kinase kinase-beta (CaMKK beta), and TGF-beta-activated kinase-1 (TAK-1), can activate AMPK by phosphorylating a threonine residue on its catalytic alpha-subunit. Once activated, AMPK leads to a concomitant inhibition of energy-consuming biosynthetic pathways, such as protein, fatty acid and glycogen synthesis, and activation of ATP-producing catabolic pathways, such as fatty acid oxidation and glycolysis. |
| map04210 | Apoptosis | Apoptosis is a genetically programmed process for the elimination of damaged or redundant cells by activation of caspases (aspartate-specific cysteine proteases). The onset of apoptosis is controlled by numerous interrelating processes. The 'extrinsic' pathway involves stimulation of members of the tumor necrosis factor (TNF) receptor subfamily, such as TNFRI, CD95/Fas or TRAILR (death receptors), located at the cell surface, by their specific ligands, such as TNF-alpha, FasL or TRAIL, respectively. The 'intrinsic' pathway is activated mainly by non-receptor stimuli, such as DNA damage, ER stress, metabolic stress, UV radiation or growth-factor deprivation. The central event in the 'intrinsic' pathway is the mitochondrial outer membrane permeabilization (MOMP), which leads to the release of cytochrome c. These two pathways converge at the level of effector caspases, such as caspase-3 and caspase-7. The third major pathway is initiated by the constituents of cytotoxic granules (e.g. Perforin and Granzyme B) that are released by CTLs (cytotoxic T-cells) and NK (natural killer) cells. Granzyme B, similarly to the caspases, cleaves its substrates after aspartic acid residues, suggesting that this protease has the ability to activate members of the caspase family directly. It is the balance between the pro-apoptotic and anti-apoptotic signals that eventually determines whether cells will undergo apoptosis, survive or proliferate. TNF family of ligands activates anti-apoptotic or cell-survival signals as well as apoptotic signals. NGF and Interleukin-3 promotes the survival, proliferation and differentiation of neurons or hematopoietic cells, respectively. Withdrawal of these growth factors leads to cell death, as described above. |
| map04211 | Longevity regulating pathway | Regulation of longevity depends on genetic and environmental factors. Caloric restriction (CR), that is limiting food intake, is recognized in mammals as the best characterized and most reproducible strategy for extending lifespan. Four pathways have been implicated in mediating the CR effect. These are the insulin like growth factor (IGF-1)/insulin signaling pathway, the sirtuin pathway, the adenosine monophosphate (AMP) activated protein kinase (AMPK) pathway and the target of rapamycin (TOR) pathway. The collective response of these pathways to CR is believed to promote cellular fitness and ultimately longevity via activation of autophagy, stress defense mechanisms, and survival pathways while attenuating proinflammatory mediators and cellular growth. Furthermore, there is evidence supporting that life span extension can be achieved with pharmacologic agents that mimic the effects of caloric restriction, such as rapamycin, via mTOR signaling blockade, resveratrol, by activating SIRT1 activity, and metformin, which seems to be a robust stimulator of AMPK activity. As an aging suppressor, Klotho is an important molecule in aging processes and its overexpression results in longevity. |
| map04212 | Longevity regulating pathway - worm | The nematode Caenorhabditis elegans has proven to be a very useful tool for studying the genetics of longevity due to its simple biology. Over 70 genes have been found to influence lifespan in this worm. The majority of the genes could be placed into canonical longevity pathways including insulin/IGF, germline, TOR signaling, and mitochondrial respiration. The insulin/IGF-1 signaling (IIS) pathway is a central regulator of life span. Transcription factors regulated by the IIS and the TOR-S6K pathways are implicated in the anti-aging effects of Dietary restriction (DR). Lifespan extension via germline removal depends on at least four signaling mechanisms: reduced TOR signaling, DAF-16/FOXO regulation, increased steroid signaling via the DAF-36/DAF-9/DAF-12 pathway, and increased NHR-80/HNF-4 signaling. The mitochondrial unfolded protein response (UPRmt), a mitochondrial stress response, can be regarded as a hormetic mechanism that extends lifespan in spite of mitochondrial dysfunction. Longevity- promoting compounds such as rapamycin and resveratrol also induce mitonuclear imbalance that activates the UPRmt. |
| map04213 | Longevity regulating pathway - multiple species | Aging is a complex process of accumulation of molecular, cellular, and organ damage, leading to loss of function and increased vulnerability to disease and death. Despite the complexity of aging, recent work has shown that dietary restriction (DR) and genetic down-regulation of nutrient-sensing pathways, namely IIS (insulin/insulin-like growth factor signalling) and TOR (target-of- rapamycin) can substantially increase healthy life span of laboratory model organisms. These nutrient signaling pathways are conserved in various organisms. In worms, flies, and mammals, DR reduces signalling through IIS/TOR pathways, deactivating the PI3K/Akt/TOR intracellular signalling cascade and consequently activating the antiaging FOXO family transcription factor(s). In yeast, the effects of DR on life- span extension are associated with reduced activities of the TOR/Sch9 and Ras/PKA pathways and require the serine-threonine kinase Rim15 and transcription factors Gis1 and Msn2/4. These transcription factors (FOXO, DAF-16, Gis1, and Msn2/4) transactivate genes involved in resistance to oxidative stress, energy metabolism, DNA damage repair, glucose metabolism, autophagy and protection of proteins by chaperones. |
| map04218 | Cellular senescence | Cellular senescence is a state of irreversible cellular arrest and can be triggered by a number of factors, such as telomere shortening, oncogene activation, irradiation, DNA damage and oxidative stress. It is characterized by enlarged flattened morphology, senescence-associated beta-galactosidase (SA-b-gal) activity, secretion of inflammatory cytokines, growth factors and matrix metalloproteinases, as part of the senescence-associated secretory phenotype (SASP). Cellular senescence is functionally associated with many biological processes including aging, tumor suppression, placental biology, embryonic development, and wound healing. |
| map04370 | VEGF signaling pathway | There is now much evidence that VEGFR-2 is the major mediator of VEGF-driven responses in endothelial cells and it is considered to be a crucial signal transducer in both physiologic and pathologic angiogenesis. The binding of VEGF to VEGFR-2 leads to a cascade of different signaling pathways, resulting in the up-regulation of genes involved in mediating the proliferation and migration of endothelial cells and promoting their survival and vascular permeability. For example, the binding of VEGF to VEGFR-2 leads to dimerization of the receptor, followed by intracellular activation of the PLCgamma;PKC-Raf kinase-MEK-mitogen-activated protein kinase (MAPK) pathway and subsequent initiation of DNA synthesis and cell growth, whereas activation of the phosphatidylinositol 3' -kinase (PI3K)-Akt pathway leads to increased endothelial-cell survival. Activation of PI3K, FAK, and p38 MAPK is implicated in cell migration signaling. |
| map04510 | Focal adhesion | Cell-matrix adhesions play essential roles in important biological processes including cell motility, cell proliferation, cell differentiation, regulation of gene expression and cell survival. At the cell-extracellular matrix contact points, specialized structures are formed and termed focal adhesions, where bundles of actin filaments are anchored to transmembrane receptors of the integrin family through a multi-molecular complex of junctional plaque proteins. Some of the constituents of focal adhesions participate in the structural link between membrane receptors and the actin cytoskeleton, while others are signalling molecules, including different protein kinases and phosphatases, their substrates, and various adapter proteins. Integrin signaling is dependent upon the non-receptor tyrosine kinase activities of the FAK and src proteins as well as the adaptor protein functions of FAK, src and Shc to initiate downstream signaling events. These signalling events culminate in reorganization of the actin cytoskeleton; a prerequisite for changes in cell shape and motility, and gene expression. Similar morphological alterations and modulation of gene expression are initiated by the binding of growth factors to their respective receptors, emphasizing the considerable crosstalk between adhesion- and growth factor-mediated signalling. |
| map04611 | Platelet activation | Platelets play a key and beneficial role for primary hemostasis on the disruption of the integrity of vessel wall. Platelet adhesion and activation at sites of vascular wall injury is initiated by adhesion to adhesive macromolecules, such as collagen and von Willebrand factor (vWF), or by soluble platelet agonists, such as ADP, thrombin, and thromboxane A2. Different receptors are stimulated by various agonists, almost converging in increasing intracellular Ca2+ concentration that stimulate platelet shape change and granule secretion and ultimately induce the inside-out signaling process leading to activation of the ligand-binding function of integrin alpha IIb beta 3. Binding of alpha IIb beta 3 to its ligands, mainly fibrinogen, mediates platelet adhesion and aggregation and triggers outside-in signaling, resulting in platelet spreading, additional granule secretion, stabilization of platelet adhesion and aggregation, and clot retraction. |
| map04650 | Natural killer cell mediated cytotoxicity | Natural killer (NK) cells are lymphocytes of the innate immune system that are involved in early defenses against both allogeneic (nonself) cells and autologous cells undergoing various forms of stress, such as infection with viruses, bacteria, or parasites or malignant transformation. Although NK cells do not express classical antigen receptors of the immunoglobulin gene family, such as the antibodies produced by B cells or the T cell receptor expressed by T cells, they are equipped with various receptors whose engagement allows them to discriminate between target and nontarget cells. Activating receptors bind ligands on the target cell surface and trigger NK cell activation and target cell lysis. However Inhibitory receptors recognize MHC class I molecules (HLA) and inhibit killing by NK cells by overruling the actions of the activating receptors. This inhibitory signal is lost when the target cells do not express MHC class I and perhaps also in cells infected with virus, which might inhibit MHC class I exprssion or alter its conformation. The mechanism of NK cell killing is the same as that used by the cytotoxic T cells generated in an adaptive immune response; cytotoxic granules are released onto the surface of the bound target cell, and the effector proteins they contain penetrate the cell membrane and induce programmed cell death. |
| map04660 | T cell receptor signaling pathway | Activation of T lymphocytes is a key event for an efficient response of the immune system. It requires the involvement of the T-cell receptor (TCR) as well as costimulatory molecules such as CD28. Engagement of these receptors through the interaction with a foreign antigen associated with major histocompatibility complex molecules and CD28 counter-receptors B7.1/B7.2, respectively, results in a series of signaling cascades. These cascades comprise an array of protein-tyrosine kinases, phosphatases, GTP-binding proteins and adaptor proteins that regulate generic and specialised functions, leading to T-cell proliferation, cytokine production and differentiation into effector cells. |
| map04662 | B cell receptor signaling pathway | B cells are an important component of adaptive immunity. They produce and secrete millions of different antibody molecules, each of which recognizes a different (foreign) antigen. The B cell receptor (BCR) is an integral membrane protein complex that is composed of two immunoglobulin (Ig) heavy chains, two Ig light chains and two heterodimers of Ig-alpha and Ig-beta. After BCR ligation by antigen, three main protein tyrosine kinases (PTKs) -the SRC-family kinase LYN, SYK and the TEC-family kinase BTK- are activated. Phosphatidylinositol 3-kinase (PI3K) and phospholipase C-gamma 2 (PLC-gamma 2) are important downstream effectors of BCR signalling. This signalling ultimately results in the expression of immediate early genes that further activate the expression of other genes involved in B cell proliferation, differentiation and Ig production as well as other processes. |
| map04664 | Fc epsilon RI signaling pathway | Fc epsilon RI-mediated signaling pathways in mast cells are initiated by the interaction of antigen (Ag) with IgE bound to the extracellular domain of the alpha chain of Fc epsilon RI. The activation pathways are regulated both positively and negatively by the interactions of numerous signaling molecules. Mast cells that are thus activated release preformed granules which contain biogenic amines (especially histamines) and proteoglycans (especially heparin). The activation of phospholipase A2 causes the release of membrane lipids followed by development of lipid mediators such as leukotrienes (LTC4, LTD4 and LTE4) and prostaglandins (especially PDG2). There is also secretion of cytokines, the most important of which are TNF-alpha, IL-4 and IL-5. These mediators and cytokines contribute to inflammatory responses. |
| map04666 | Fc gamma R-mediated phagocytosis | Phagocytosis plays an essential role in host-defense mechanisms through the uptake and destruction of infectious pathogens. Specialized cell types including macrophages, neutrophils, and monocytes take part in this process in higher organisms. After opsonization with antibodies (IgG), foreign extracellular materials are recognized by Fc gamma receptors. Cross-linking of Fc gamma receptors initiates a variety of signals mediated by tyrosine phosphorylation of multiple proteins, which lead through the actin cytoskeleton rearrangements and membrane remodeling to the formation of phagosomes. Nascent phagosomes undergo a process of maturation that involves fusion with lysosomes. The acquisition of lysosomal proteases and release of reactive oxygen species are crucial for digestion of engulfed materials in phagosomes. |
| map04722 | Neurotrophin signaling pathway | Neurotrophins are a family of trophic factors involved in differentiation and survival of neural cells. The neurotrophin family consists of nerve growth factor (NGF), brain derived neurotrophic factor (BDNF), neurotrophin 3 (NT-3), and neurotrophin 4 (NT-4). Neurotrophins exert their functions through engagement of Trk tyrosine kinase receptors or p75 neurotrophin receptor (p75NTR). Neurotrophin/Trk signaling is regulated by connecting a variety of intracellular signaling cascades, which include MAPK pathway, PI-3 kinase pathway, and PLC pathway, transmitting positive signals like enhanced survival and growth. On the other hand, p75NTR transmits both positive and nagative signals. These signals play an important role for neural development and additional higher-order activities such as learning and memory. |
| map04810 | Regulation of actin cytoskeleton | NA |
| map04910 | Insulin signaling pathway | Insulin binding to its receptor results in the tyrosine phosphorylation of insulin receptor substrates (IRS) by the insulin receptor tyrosine kinase (INSR). This allows association of IRSs with the regulatory subunit of phosphoinositide 3-kinase (PI3K). PI3K activates 3-phosphoinositide-dependent protein kinase 1 (PDK1), which activates Akt, a serine kinase. Akt in turn deactivates glycogen synthase kinase 3 (GSK-3), leading to activation of glycogen synthase (GYS) and thus glycogen synthesis. Activation of Akt also results in the translocation of GLUT4 vesicles from their intracellular pool to the plasma membrane, where they allow uptake of glucose into the cell. Akt also leads to mTOR-mediated activation of protein synthesis by eIF4 and p70S6K. The translocation of GLUT4 protein is also elicited through the CAP/Cbl/TC10 pathway, once Cbl is phosphorylated by INSR.Other signal transduction proteins interact with IRS including GRB2. GRB2 is part of the cascade including SOS, RAS, RAF and MEK that leads to activation of mitogen-activated protein kinase (MAPK) and mitogenic responses in the form of gene transcription. SHC is another substrate of INSR. When tyrosine phosphorylated, SHC associates with GRB2 and can thus activate the RAS/MAPK pathway independently of IRS-1. |
| map04914 | Progesterone-mediated oocyte maturation | Xenopus oocytes are naturally arrested at G2 of meiosis I. Exposure to either insulin/IGF-1 or the steroid hormone progesterone breaks this arrest and induces resumption of the two meiotic division cycles and maturation of the oocyte into a mature, fertilizable egg. This process is termed oocyte maturation. The transition is accompanied by an increase in maturation promoting factor (MPF or Cdc2/cyclin B) which precedes germinal vesicle breakdown (GVBD). Most reports point towards the Mos-MEK1-ERK2 pathway [where ERK is an extracellular signal-related protein kinase, MEK is a MAPK/ERK kinase and Mos is a p42(MAPK) activator] and the polo-like kinase/CDC25 pathway as responsible for the activation of MPF in meiosis, most likely triggered by a decrease in cAMP. |
| map04917 | Prolactin signaling pathway | Prolactin (PRL) is a polypeptide hormone known to be involved in a wide range of biological functions including osmoregulation, lactation, reproduction, growth and development, endocrinology and metabolism, brain and behavior, and immunomodulation. PRL mediates its action through PRLR, a transmembrane protein of the hematopoietin cytokine receptor superfamily. At the protein level, the long PRLR isoform (long-R) and several short PRLR isoforms (short-R) have been detected. Acting through the long-R, PRL activates many signaling cascades including Jak2/Stat, the major cascade, Src kinase, phosphatidylinositol-3-kinase (PI3K)/AKT, and mitogen-activated protein kinase (MAPK) pathways. PRL cannot activate Jak2/Stat5 through the short-R, but can activate pathways including MAPK and PI3K pathways. |
| map04919 | Thyroid hormone signaling pathway | The thyroid hormones (THs) are important regulators of growth, development and metabolism. The action of TH is mainly mediated by T3 (3,5,3'-triiodo-L-thyronine). Thyroid hormones, L-thyroxine (T4) and T3 enter the cell through transporter proteins. Although the major form of TH in the blood is T4, it is converted to the more active hormone T3 within cells. T3 binds to nuclear thyroid hormone receptors (TRs), which functions as a ligand-dependent transcription factor and controls the expression of target genes (genomic action). Nongenomic mechanisms of action is initiated at the integrin receptor. The plasma membrane alpha(v)beta(3)-integrin has distinct binding sites for T3 and T4. One binding site binds only T3 and activates the phosphatidylinositol 3-kinase (PI3K) pathway. The other binding site binds both T3 and T4 and activates the ERK1/2 MAP kinase pathway. |
| map04926 | Relaxin signaling pathway | Human relaxin-2 (relaxin), originally identified as a peptidic hormone of pregnancy, is now known to exert a range of pleiotropic effects including vasodilatory, anti-fibrotic and angiogenic effects in both males and females. It belongs to the so-called relaxin peptide family which includes the insulin-like peptides INSL3 and INSL5, and relaxin-3 (H3) as well as relaxin. INSL3 has clearly defined specialist roles in male and female reproduction, relaxin-3 is primarily a neuropeptide involved in stress and metabolic control, and INSL5 is widely distributed particularly in the gastrointestinal tract. These members of relaxin peptide family exert such effects binding to different kinds of receptors, classified as relaxin family peptide (RXFP) receptors: RXFP1, RXFP2, RXFP3, and RXFP4. These G protein-coupled receptors predominantly bind relaxin, INSL3, relaxin-3, and INSL-5, respectively. RXFP1 activates a wide spectrum of signaling pathways to generate second messengers that include cAMP and nitric oxide, whereas RXFP2 activates a subset of these pathways. Both RXFP3 and RXFP4 inhibit cAMP production, and RXFP3 activate MAP kinases. |
| map04931 | Insulin resistance | Insulin resistance is a condition where cells become resistant to the effects of insulin. It is often found in people with health disorders, including obesity, type 2 diabetes mellitus, non-alcoholic fatty liver disease, and cardiovascular diseases. In this diagram multiple mechanisms underlying insulin resistance are shown: (a) increased phosphorylation of IRS (insulin receptor substrate) protein through serine/threonine kinases, such as JNK1 and IKKB, and protein kinase C, (b) increased IRS-1 proteasome degradation via mTOR signaling pathway, (c) decreased activation of signaling molecules including PI3K and AKT, (d) increase in activity of phosphatases including PTPs, PTEN, and PP2A. Regulatory actions such as oxidative stress, mitochondrial dysfunction, accumulation of intracellular lipid derivatives (diacylglycrol and ceramides), and inflammation (via IL-6 and TNFA) contribute to these mechanisms. Consequently, insulin resistance causes reduced GLUT4 translocation, resulting in glucose takeup and glycogen synthesis in skeletal muscle as well as increased hepatic gluconeogenesis and decreased glycogen synthesis in liver. At the bottom of the diagram, interplay between O-GlcNAcylation and serine/threonine phosphorylation is shown. Studies suggested that elevated O-GlcNAc level was correlated to high glucose-induced insulin resistance. Donor UDP-GlcNAc is induced through hexosamine biosynthesis pathway and added to proteins by O-GlcNAc transferase. Elevation of O-GlcNAc modification alters phosphorylation and function of key insulin signaling proteins including IRS-1, PI3K, PDK1, Akt and other transcription factor and cofactors, resulting in the attenuation of insulin signaling cascade. |
| map04960 | Aldosterone-regulated sodium reabsorption | Sodium transport across the tight epithelia of Na+ reabsorbing tissues such as the distal part of the kidney nephron and colon is the major factor determining total-body Na+ levels, and thus, long-term blood pressure. Aldosterone plays a major role in sodium and potassium metabolism by binding to epithelial mineralocorticoid receptors (MR) in the renal collecting duct cells localized in the distal nephron, promoting sodium resorption and potassium excretion. Aldosterone enters a target cell and binds MR, which translocates into the nucleus and regulates gene transcription. Activation of MR leads to increased expression of Sgk-1, which phosphorylates Nedd4-2, an ubiquitin-ligase which targets ENAC to proteosomal degradation. Phosphorylated Nedd4-2 dissociates from ENAC, increasing its apical membrane abundance. Activation of MR also leads to increased expression of Na+/K+-ATPase, thus causing a net increase in sodium uptake from the renal filtrate. The specificity of MR for aldosterone is provided by 11beta-HSD2 by the rapid conversion of cortisol to cortisone in renal cortical collecting duct cells. Recently, besides genomic effects mediated by activated MR, rapid aldosterone actions that are independent of translation and transcription have been documented. |
| map04973 | Carbohydrate digestion and absorption | Dietary carbohydrate in humans and omnivorous animals is a major nutrient. The carbohydrates that we ingest vary from the lactose in milk to complex carbohydrates. These carbohydrates are digested to monosaccharides, mostly glucose, galactose and fructose, prior to absorption in the small intestine. Glucose and galactose are initially transported into the enterocyte by SGLT1 located in the apical brush border membrane and then exit through the basolateral membrane by either GLUT2 or exocytosis. In a new model of intestinal glucose absorption, transport by SGLT1 induces rapid insertion and activation of GLUT2 in the brush border membrane by a PKC betaII-dependent mechanism. Moreover, trafficking of apical GLUT2 is rapidly up-regulated by glucose and artificial sweeteners, which act through T1R2 + T1R3/alpha-gustducin to activate PLC-beta2 and PKC-beta II. Fructose is transported separately by the brush border GLUT5 and then released out of the enterocyte into the blood by GLUT2. |
| map05142 | Chagas disease (American trypanosomiasis) | Trypanosoma cruzi is an intracellular protozoan parasite that causes Chagas disease. The parasite life cycle involves hematophagous reduviid bugs as vectors. Once parasites enter the host body, they invade diverse host cells including cardiomyocytes. Establishment of infection depends on various parasite molecules such as cruzipain, oligopeptidase B, and trans-sialidase that activate Ca2+ signaling. Internalized parasites escape from the parasitophorous vacuole using secreted pore-forming TcTOX molecule and replicate in the cytosol. Multiplied parasites eventually lyse infected host cells and are released in the circulation. During these events, the parasites manipulate host innate immunity and elicit cardiomyocyte hypertrophy. T lymphocyte responses are also disturbed. |
| map05145 | Toxoplasmosis | Toxoplasma gondii is an obligate intracellular parasite that is prevalent worldwide. The tachyzoite form acquired by oral ingestion downmodulates proinflammatory signaling pathways via various mechanisms. During early infection, nuclear translocation of NFkB is temporally blocked and p38 MAPK phosphorylation is prevented, suppressing IL-12 production. Another pathway for IL-12 induction occurs through CCR5 dependent pathway, but parasitic induction of an eicosanoid LXA4 contributes to the downregulation of IL-12. Direct activation of STAT3 by the parasite enhance anti-inflammatory function of IL-10 and TGF beta. T. gondii can cause lifelong chronic infection by establishing an anti-apoptotic environment through induction of bcl-2 or IAPs and by redirecting LDL-mediated cholesterol transport to scavenge nutrients from the host. |
| map05160 | Hepatitis C | Hepatitis C virus (HCV) is a major cause of chronic liver disease. The HCV employ several strategies to perturb host cell immunity. After invasion, HCV RNA genome functions directly as an mRNA in the cytoplasm of the host cell and forms membrane-associated replication complexes along with non-structural proteins. Viral RNA can trigger the RIG-I pathway and interferon production during this process. Translated HCV protein products regulate immune response to inhibit the action of interferon. HCV core and NS5A proteins appear to be the most important molecules with regulatory functions that modulate transcription, cellular proliferation, and apoptosis. |
| map05162 | Measles | Measles virus (MV) is highly contagious virus that leads infant death worldwide. Humans are the unique natural reservoir for this virus. It causes severe immunosuppression favouring secondary bacterial infections. Several MV proteins have been suggested to disturb host immunity. After infection of host lymphoid cells via SLAM, MV inhibits cytokine response by direct interference with host signaling systems. Three proteins (P, V, and C) associate with Jak/STAT proteins in interferon-triggered pathway and other important proteins related to apoptosis. Interaction between MV and host brings about the shift towards a Th2 response by decreasing IL-12 production and induces lymphopenia by suppressing cell proliferation. |
| map05163 | Human cytomegalovirus infection | Human cytomegalovirus (HCMV) is an enveloped, double-stranded DNA virus that is a member of beta-herpesvirus family. HCMV is best known for causing significant morbidity and mortality in immunocompromised populations. As with other herpesviruses, HCMV gB and gH/gL envelope glycoproteins are essential for virus entry. HCMV gB could activate the PDGFRA, and induce activation of the oncogenic PI3-K/AKT pathway. Though it is unlikely that HCMV by itself can act as an oncogenic factor, HCMV may have an oncomodulatory role, to catalyze an oncogenic process that has already been initiated. US28, one of the four HCMV-encoded vGPCRs (US27, US28, UL33 and UL78), also has a specific role in the oncomodulatory properties. In addition, HCMV has developed numerous mechanisms for manipulating the host immune system. The virally encoded US2, US3, US6 and US11 gene products all interfere with major histocompatibility complex (MHC) class I antigen presentation. HCMV encodes several immediate early (IE) antiapoptotic proteins (IE1, IE2, vMIA and vICA). These proteins might avoid immune clearance of infected tumor cells by cytotoxic lymphocytes and NK cells. |
| map05164 | Influenza A | Influenza is a contagious respiratory disease caused by influenza virus infection. Influenza A virus is responsible for both annual seasonal epidemics and periodic worldwide pandemics. Novel strains that cause pandemics arise from avian influenza virus by genetic reassortment among influenza viruses and two surface glycoproteins HA and NA form the basis of serologically distinct virus types. The innate immune system recognizes invaded virus through multiple mechanisms. Viral non-structural NS1 protein is a multifunctional virulence factor that interfere IFN-mediated antiviral response. It inhibits IFN production by blocking activation of transcription factors such as NF-kappa B, IRF3 and AP1. NS1 further inhibits the activation of IFN-induced antiviral genes. PB1-F2 protein is another virulence factor that induce apoptosis of infected cells, which results in life-threatening bronchiolitis. |
| map05165 | Human papillomavirus infection | Human papillomavirus (HPV) is a non-enveloped, double-stranded DNA virus. HPV infect mucoal and cutaneous epithelium resulting in several types of pathologies, most notably, cervical cancer. All types of HPV share a common genomic structure and encode eight proteins: E1, E2, E4, E5, E6, and E7 (early) and L1 and L2 (late). It has been demonstrated that E1 and E2 are involved in viral transcription and replication. The functions of the E4 protein is not yet fully understood. E5, E6, and E7 act as oncoproteins. E5 inhibits the V-ATPase, prolonging EGFR signaling and thereby promoting cell proliferation. The expression of E6 and E7 not only inhibits the tumor suppressors p53 and Rb, but also alters additional signalling pathways. Among these pathways, PI3K/Akt signalling cascade plays a very important role in HPV-induced carcinogenesis. The L1 and L2 proteins form icosahedral capsids for progeny virion generation. |
| map05166 | Human T-cell leukemia virus 1 infection | Human T-cell leukemia virus type 1 (HTLV-1) is a pathogenic retrovirus that is associated with adult T-cell leukemia/lymphoma (ATL). It is also strongly implicated in non-neoplastic chronic inflammatory diseases such as HTLV-1-associated myelopathy/tropical spastic paraparesis (HAM/TSP). Expression of Tax, a viral regulatory protein is critical to the pathogenesis. Tax is a transcriptional co-factor that interfere several signaling pathways related to anti-apoptosis or cell proliferation. The modulation of the signaling by Tax involve its binding to transcription factors like CREB/ATF, NF-kappa B, and SRF. |
| map05167 | Kaposi sarcoma-associated herpesvirus infection | Kaposi sarcoma-associated herpesvirus (KSHV), also known as human herpesvirus 8 (HHV-8), is the most recently identified human tumor virus, and is associated with the pathogenesis of Kaposi's sarcoma (KS), primary effusion lymphoma (PEL), and Multicentric Castleman's disease (MCD). Like all other herpesviruses, KSHV displays two modes of life cycle, latency and lytic replication, which are characterized by the patterns of viral gene expression. Genes expressed in latency (LANA, v-cyclin, v-FLIP, Kaposins A, B and C and viral miRNAs) are mainly thought to facilitate the establishment of life long latency in its host and survival against the host innate, and adaptive immune surveillance mechanisms. Among the viral proteins shown to be expressed during lytic replication are potent signaling molecules such as vGPCR, vIL6, vIRFs, vCCLs, K1 and K15, which have been implicated experimentally in the angiogenic and inflammatory phenotype observed in KS lesions. Several of these latent viral and lytic proteins are known to transform host cells, linking KSHV with the development of severe human malignancies. |
| map05169 | Epstein-Barr virus infection | Epstein-Barr virus (EBV) is a gamma-herpes virus that widely infects human populations predominantly at an early age but remains mostly asymptomatic. EBV has been linked to a wide spectrum of human malignancies, including nasopharyngeal carcinoma and other hematologic cancers, like Hodgkin's lymphoma, Burkitt's lymphoma (BL), B-cell immunoblastic lymphoma in HIV patients, and posttransplant-associated lymphoproliferative diseases. EBV has the unique ability to establish life-long latent infection in primary human B lymphocytes. During latent infection, EBV expresses a small subset of genes, including 6 nuclear antigens (EBNA-1, -2, -3A, -3B, -3C, and -LP), 3 latent membrane proteins (LMP-1, -2A, and -2B), 2 small noncoding RNAs (EBER-1 and 2). On the basis of these latent gene expression, three different latency patterns associated with the types of cancers are recognized. |
| map05170 | Human immunodeficiency virus 1 infection | Human immunodeficiency virus type 1 (HIV-1) , the causative agent of AIDS (acquired immunodeficiency syndrome), is a lentivirus belonging to the Retroviridae family. The primary cell surface receptor for HIV-1, the CD4 protein, and the co-receptor for HIV-1, either CCR5 or CXCR4, are found on macrophages and T lymphocytes. At the earliest step, sequential binding of virus envelope (Env) glycoprotein gp120 to CD4 and the co-receptor CCR5 or CXCR4 facilitates HIV-1 entry and has the potential to trigger critical signaling that may favor viral replication. At advanced stages of the disease, HIV-1 infection results in dramatic induction of T-cell (CD4+ T and CD8+ T cell) apoptosis both in infected and uninfected bystander T cells, a hallmark of HIV-1 pathogenesis. On the contrary, macrophages are resistant to the cytopathic effect of HIV-1 and produce virus for longer periods of time. |
| map05200 | Pathways in cancer | NA |
| map05210 | Colorectal cancer | Colorectal cancer (CRC) is the second largest cause of cancer-related deaths in Western countries. CRC arises from the colorectal epithelium as a result of the accumulation of genetic alterations in defined oncogenes and tumour suppressor genes (TSG). Two major mechanisms of genomic instability have been identified in sporadic CRC progression. The first, known as chromosomal instability (CIN), results from a series of genetic changes that involve the activation of oncogenes such as K-ras and inactivation of TSG such as p53, DCC/Smad4, and APC. The second, known as microsatellite instability (MSI), results from inactivation of the DNA mismatch repair genes MLH1 and/or MSH2 by hypermethylation of their promoter, and secondary mutation of genes with coding microsatellites, such as transforming growth factor receptor II (TGF-RII) and BAX. Hereditary syndromes have germline mutations in specific genes (mutation in the tumour suppressor gene APC on chromosome 5q in FAP, mutated DNA mismatch repair genes in HNPCC). |
| map05212 | Pancreatic cancer | Infiltrating ductal adenocarcinoma is the most common malignancy of the pancreas. When most investigators use the term 'pancreatic cancer' they are referring to pancreatic ductal adenocarcinoma (PDA). Normal duct epithelium progresses to infiltrating cancer through a series of histologically defined precursors (PanINs). The overexpression of HER-2/neu and activating point mutations in the K-ras gene occur early, inactivation of the p16 gene at an intermediate stage, and the inactivation of p53, SMAD4, and BRCA2 occur relatively late. Activated K-ras engages multiple effector pathways. Although EGF receptors are conventionally regarded as upstream activators of RAS proteins, they can also act as RAS signal transducers via RAS-induced autocrine activation of the EGFR family ligands. Moreover, PDA shows extensive genomic instability and aneuploidy. Telomere attrition and mutations in p53 and BRCA2 are likely to contribute to these phenotypes. Inactivation of the SMAD4 tumour suppressor gene leads to loss of the inhibitory influence of the transforming growth factor-beta signalling pathway. |
| map05213 | Endometrial cancer | Endometrial cancer (EC) is the most common gynaecological malignancy and the fourth most common malignancy in women in the developed world after breast, colorectal and lung cancer. Two types of endometrial carcinoma are distinguished with respect to biology and clinical course. Type-I carcinoma is related to hyperestrogenism by association with endometrial hyperplasia, frequent expression of estrogen and progesterone receptors and younger age, whereas type-II carcinoma is unrelated to estrogen, associated with atrophic endometrium, frequent lack of estrogen and progesterone receptors and older age. The morphologic differences in these cancers are mirrored in their molecular genetic profile with type I showing defects in DNA-mismatch repair and mutations in PTEN, K-ras, and beta-catenin, and type II showing aneuploidy, p53 mutations, and her2/neu amplification. |
| map05214 | Glioma | Gliomas are the most common of the primary brain tumors and account for more than 40% of all central nervous system neoplasms. Gliomas include tumours that are composed predominantly of astrocytes (astrocytomas), oligodendrocytes (oligodendrogliomas), mixtures of various glial cells (for example,oligoastrocytomas) and ependymal cells (ependymomas). The most malignant form of infiltrating astrocytoma - glioblastoma multiforme (GBM) - is one of the most aggressive human cancers. GBM may develop de novo (primary glioblastoma) or by progression from low-grade or anaplastic astrocytoma (secondary glioblastoma). Primary glioblastomas develop in older patients and typically show genetic alterations (EGFR amplification, p16/INK4a deletion, and PTEN mutations) at frequencies of 24-34%. Secondary glioblastomas develop in younger patients and frequently show overexpression of PDGF and CDK4 as well as p53 mutations (65%) and loss of Rb playing major roles in such transformations. Loss of PTEN has been implicated in both pathways, although it is much more common in the pathogenesis of primary GBM. |
| map05215 | Prostate cancer | Prostate cancer constitutes a major health problem in Western countries. It is the most frequently diagnosed cancer among men and the second leading cause of male cancer deaths. The identification of key molecular alterations in prostate-cancer cells implicates carcinogen defenses (GSTP1), growth-factor-signaling pathways (NKX3.1, PTEN, and p27), and androgens (AR) as critical determinants of the phenotype of prostate-cancer cells. Glutathione S-transferases (GSTP1) are detoxifying enzymes. Cells of prostatic intraepithelial neoplasia, devoid of GSTP1, undergo genomic damage mediated by carcinogens. NKX3.1, PTEN, and p27 regulate the growth and survival of prostate cells in the normal prostate. Inadequate levels of PTEN and NKX3.1 lead to a reduction in p27 levels and to increased proliferation and decreased apoptosis. Androgen receptor (AR) is a transcription factor that is normally activated by its androgen ligand. During androgen withdrawal therapy, the AR signal transduction pathway also could be activated by amplification of the AR gene, by AR gene mutations, or by altered activity of AR coactivators. Through these mechanisms, tumor cells lead to the emergence of androgen-independent prostate cancer. |
| map05218 | Melanoma | Melanoma is a form of skin cancer that has a poor prognosis and which is on the rise in Western populations. Melanoma arises from the malignant transformation of pigment-producing cells, melanocytes. The only known environmental risk factor is exposure to ultraviolet (UV) light and in people with fair skin the risk is greatly increased. Melanoma pathogenesis is also driven by genetic factors. Oncogenic NRAS mutations activate both effector pathways Raf-MEK-ERK and PI3K-Akt. The Raf-MEK-ERK pathway may also be activated via mutations in the BRAF gene. The PI3K-Akt pathway may be activated through loss or mutation of the inhibitory tumor suppressor gene PTEN. These mutations arise early during melanoma pathogenesis and are preserved throughout tumor progression. Melanoma development has been shown to be strongly associated with inactivation of the p16INK4a/cyclin dependent kinases 4 and 6/retinoblastoma protein (p16INK4a/CDK4,6/pRb) and p14ARF/human double minute 2/p53 (p14ARF/HMD2/p53) tumor suppressor pathways. MITF and TP53 are implicated in further melanoma progression. |
| map05220 | Chronic myeloid leukemia | Chronic myeloid leukemia (CML) is a clonal myeloproliferative disorder of a pluripotent stem cell. The natural history of CML has a triphasic clinical course comprising of an initial chronic phase (CP), which is characterized by expansion of functionally normal myeloid cells, followed by an accelerated phase (AP) and finally a more aggressive blast phase (BP), with loss of terminal differentiation capacity. On the cellular level, CML is associated with a specific chromosome abnormality, the t(9; 22) reciprocal translocation that forms the Philadelphia (Ph) chromosome. The Ph chromosome is the result of a molecular rearrangement between the c-ABL proto-oncogene on chromosome 9 and the BCR (breakpoint cluster region) gene on chromosome 22. The BCR/ABL fusion gene encodes p210 BCR/ABL, an oncoprotein, which, unlike the normal p145 c-Abl, has constitutive tyrosine kinase activity and is predominantly localized in the cytoplasm. While fusion of c-ABL and BCR is believed to be the primary cause of the chronic phase of CML, progression to blast crisis requires other molecular changes. Common secondary abnormalities include mutations in TP53, RB, and p16/INK4A, or overexpression of genes such as EVI1. Additional chromosome translocations are also observed,such as t(3;21)(q26;q22), which generates AML1-EVI1. |
| map05221 | Acute myeloid leukemia | Acute myeloid leukemia (AML) is a disease that is characterized by uncontrolled proliferation of clonal neoplastic cells and accumulation in the bone marrow of blasts with an impaired differentiation program. AML accounts for approximately 80% of all adult leukemias and remains the most common cause of leukemia death. Two major types of genetic events have been described that are crucial for leukemic transformation. A proposed necessary first event is disordered cell growth and upregulation of cell survival genes. The most common of these activating events were observed in the RTK Flt3, in N-Ras and K-Ras, in Kit, and sporadically in other RTKs. Alterations in myeloid transcription factors governing hematopoietic differentiation provide second necessary event for leukemogenesis. Transcription factor fusion proteins such as AML-ETO, PML-RARalpha or PLZF-RARalpha block myeloid cell differentiation by repressing target genes. In other cases, the transcription factors themselves are mutated. |
| map05222 | Small cell lung cancer | Lung cancer is a leading cause of cancer death among men and women in industrialized countries. Small cell lung carcinoma (SCLC) is a highly aggressive neoplasm, which accounts for approximately 25% of all lung cancer cases. Molecular mechanisms altered in SCLC include induced expression of oncogene, MYC, and loss of tumorsuppressor genes, such as p53, PTEN, RB, and FHIT. The overexpression of MYC proteins in SCLC is largely a result of gene amplification. Such overexpression leads to more rapid proliferation and loss of terminal differentiation. Mutation or deletion of p53 or PTEN can lead to more rapid proliferation and reduced apoptosis. The retinoblastoma gene RB1 encodes a nuclear phosphoprotein that helps to regulate cell-cycle progression. The fragile histidine triad gene FHIT encodes the enzyme diadenosine triphosphate hydrolase, which is thought to have an indirect role in proapoptosis and cell-cycle control. |
| map05223 | Non-small cell lung cancer | Lung cancer is a leading cause of cancer death among men and women in industrialized countries. Non-small-cell lung cancer (NSCLC) accounts for approximately 85% of lung cancer and represents a heterogeneous group of cancers, consisting mainly of squamous cell (SCC), adeno (AC) and large-cell carcinoma. Molecular mechanisms altered in NSCLC include activation of oncogenes, such as K-RAS, EGFR and EML4-ALK, and inactivation of tumorsuppressor genes, such as p53, p16INK4a, RAR-beta, and RASSF1. Point mutations within the K-RAS gene inactivate GTPase activity and the p21-RAS protein continuously transmits growth signals to the nucleus. Mutations or overexpression of EGFR leads to a proliferative advantage. EML4-ALK fusion leads to constitutive ALK activation, which causes cell proliferation, invasion, and inhibition of apoptosis. Inactivating mutation of p53 can lead to more rapid proliferation and reduced apoptosis. The protein encoded by the p16INK4a inhibits formation of CDK-cyclin-D complexes by competitive binding of CDK4 and CDK6. Loss of p16INK4a expression is a common feature of NSCLC. RAR-beta is a nuclear receptor that bears vitamin-A-dependent transcriptional activity. RASSF1A is able to form heterodimers with Nore-1, an RAS effector.Therefore loss of RASSF1A might shift the balance of RAS activity towards a growth-promoting effect. |
| map05224 | Breast cancer | Breast cancer is the leading cause of cancer death among women worldwide. The vast majority of breast cancers are carcinomas that originate from cells lining the milk-forming ducts of the mammary gland. The molecular subtypes of breast cancer, which are based on the presence or absence of hormone receptors (estrogen and progesterone subtypes) and human epidermal growth factor receptor-2 (HER2), include: hormone receptor positive and HER2 negative (luminal A subtype), hormone receptor positive and HER2 positive (luminal B subtype), hormone receptor negative and HER2 positive (HER2 positive), and hormone receptor negative and HER2 negative (basal-like or triple-negative breast cancers (TNBCs)). Hormone receptor positive breast cancers are largely driven by the estrogen/ER pathway. In HER2 positive breast tumours, HER2 activates the PI3K/AKT and the RAS/RAF/MAPK pathways, and stimulate cell growth, survival and differentiation. In patients suffering from TNBC, the deregulation of various signalling pathways (Notch and Wnt/beta-catenin), EGFR protein have been confirmed. In the case of breast cancer only 8% of all cancers are hereditary, a phenomenon linked to genetic changes in BRCA1 or BRCA2. Somatic mutations in only three genes (TP53, PIK3CA and GATA3) occurred at >10% incidence across all breast cancers. |
| map05225 | Hepatocellular carcinoma | Hepatocellular carcinoma (HCC) is a major type of primary liver cancer and one of the rare human neoplasms etiologically linked to viral factors. It has been shown that, after HBV/HCV infection and alcohol or aflatoxin B1 exposure, genetic and epigenetic changes occur. The recurrent mutated genes were found to be highly enriched in multiple key driver signaling processes, including telomere maintenance, TP53, cell cycle regulation, the Wnt/beta-catenin pathway (CTNNB1 and AXIN1), the phosphatidylinositol-3 kinase (PI3K)/AKT/mammalian target of rapamycin (mTOR) pathway. Recent studies using whole-exome sequencing have revealed recurrent mutations in new driver genes involved in the chromatin remodelling (ARID1A and ARID2) and the oxidative stress (NFE2L2) pathways. |
| map05226 | Gastric cancer | Gastric cancer (GC) is one of the world's most common cancers. According to Lauren's histological classification gastric cancer is divided into two distinct histological groups - the intestinal and diffuse types. Several genetic changes have been identified in intestinal-type GC. The intestinal metaplasia is characterized by mutations in p53 gene, reduced expression of retinoic acid receptor beta (RAR-beta) and hTERT expression. Gastric adenomas furthermore display mutations in the APC gene, reduced p27 expression and cyclin E amplification. In addition, amplification and overexpression of c-ErbB2, reduced TGF-beta receptor type I (TGFBRI) expression and complete loss of p27 expression are commonly observed in more advanced GC. The main molecular changes observed in diffuse-type GCs include loss of E-cadherin function by mutations in CDH1 and amplification of MET and FGFR2F. |
| map05231 | Choline metabolism in cancer | Abnormal choline metabolism is emerging as a metabolic hallmark that is associated with oncogenesis and tumour progression. Following transformation, oncogenic signalling via pathways such as the RAS and PI3K-AKT pathways, and transcription factors associated with oncogenesis such as hypoxia-inducible factor 1 (HIF1) mediate overexpression and activation of choline cycle enzymes, which causes increased levels of choline-containing precursors and breakdown products of membrane phospholipids. These products of choline phospholipid metabolism, such as phosphocholine (PCho), diacylglycerol (DAG) and phosphatidic acid, may function as second messengers that are essential for the mitogenic activity of growth factors, particularly in the activation of the ras-raf-1-MAPK cascade and protein kinase C pathway. |

